Our body is full of invisible switches. On the surface of every cell, millions of tiny receptors pick up signals from the environment and pass them on. If these switches do not work properly, diseases can develop – from cancer to neurological disorders. A research team at Leipzig University has now deciphered a key mechanism in a special group of these receptors.
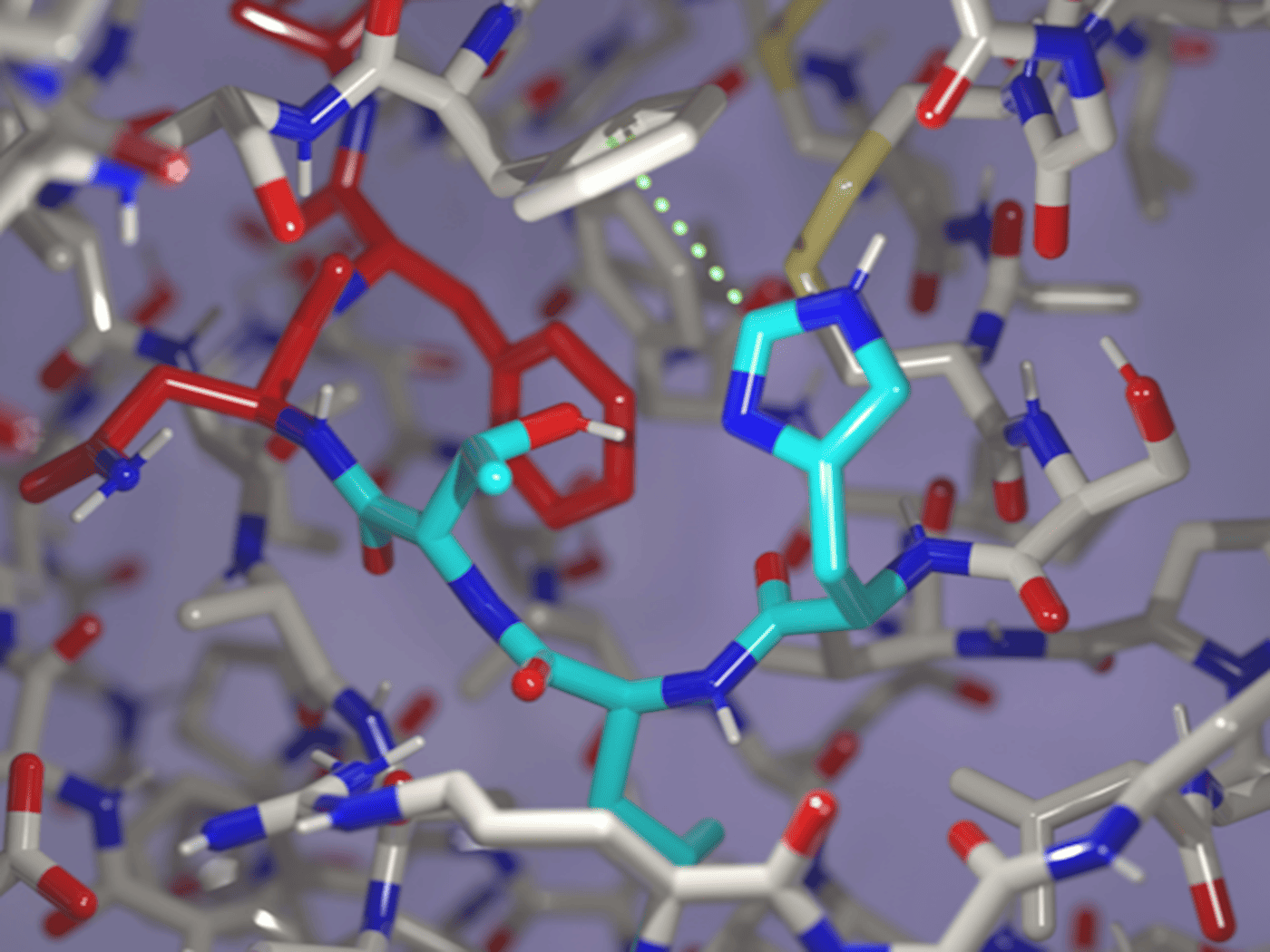
The focus is on so-called adhesion G protein-coupled receptors (aGPCRs). These protein structures sit on the cell surface and measure mechanical forces, for example when cells come into contact with each other. They belong to a family of proteins targeted by around a third of all approved drugs, yet they themselves have hardly been explored for therapeutic use so far. Their special feature: they can cleave themselves. In this process, a section of the protein splits into two parts – a step that activates the receptor. However, it has remained unclear why this self-cleavage barely occurs in some receptors.
New treatment approaches possible
Based on these findings, the researchers were able to intervene directly. They modified flexible regions of the protein and added the missing amino acid. As a result, cleavage occurred in less than two days instead of 100. “Our results show that the cleavage of adhesion GPCRs depends not only on the chemical sequence, but also on the protein’s structure and dynamics,” says Prof. Dr. Norbert Sträter from the Institute of Bioanalytical Chemistry. This is an important step toward understanding how these receptors are activated – and how they can be specifically targeted.
At the same time, the study showed that some of these receptors can function even without cleavage. In the long term, these findings could open up new treatment approaches for cancer, inflammatory diseases, and disorders of the nervous system.
Original publication:
Pohl, F., Seufert, F., Chung, Y.K. et al. Structural basis of GAIN domain autoproteolysis and cleavage-resistance in the adhesion G-protein coupled receptors. Nat Commun 17, 3259 (2026).